Density of air at different temperatures formula. Influence of air humidity. Temperature, pressure and density
Bred differential equations(1.2, 1.4) contain parameters that characterize a liquid or gas: density r , viscosity m , as well as the parameters porous medium– porosity coefficients m and permeability k . For further calculations, it is necessary to know the dependence of these coefficients on pressure.
Dropping Liquid Density. With steady filtration of a dropping liquid, its density can be considered independent of pressure, i.e., the liquid can be considered as incompressible: r = const .
Pressure increases with temperature because the particles have more kinetic energy. Imagine a box full of bouncing balls, if these balls start to move faster, the balls will hit the walls of the box harder, giving more power to the box. Pressure is just force per area, so if the force increases but the box stays the same size, the pressure increases.
The density of air can decrease with temperature if the pressure also decreases. If the pressure is constant, this cannot happen. Whenever you specify a relationship between any two pressures, density, or temperature, you must keep the third constant or specify its behavior.
In transient processes, it is necessary to take into account the compressibility of the liquid, which is characterized by liquid volumetric compression ratio b . This coefficient is usually considered constant:
Integrating the last equality from the initial pressure values p 0 and density r0 to the current values, we get:
For example, hot air rises, but why is it cold on top of the mountain. The answer is that hot air is less dense than cold air surrounding it for constant pressure, and less dense it rises. With a mountain the pressure decreases, and we also find in the atmosphere that the temperature decreases as the pressure decreases.
What usually happens on a hot day is that the surface that is heated by the sun heats the lowest level of the atmosphere, reducing its density. This will eventually lead to convection and mix this warm air vertically. Given enough time, this will reduce the mass in the air column and therefore reduce the surface pressure. They are called "thermal lows" and you can see them forming in desert areas and they play a role in sea breeze formation and monsoons.
In this case, we obtain a linear dependence of density on pressure.
Density of gases. Compressible liquids (gases) with small changes in pressure and temperature can also be characterized by volumetric compression and thermal expansion coefficients. But with large changes in pressure and temperature, these coefficients change within wide limits, so the dependence of the density of an ideal gas on pressure and temperature is based on Claiperon–Mendeleev equations of state:
To solve an extended question. At flight level, we fly on surfaces of constant pressure, which we then translate into altitude. In any given column of atmosphere, if it is warmer than standard, the given pressure surface will be higher, and if it is colder than standard, the pressure will be lower.
It follows the next question. The aneroid plates detect changes in pressure and your altimeter displays an uncorrected altitude for temperature. This is why your true altitude may vary with temperature for a constant reported altitude. When you adjust the temperature height, we call this "density height".
where R' = R/M m is the gas constant, which depends on the composition of the gas.
The gas constant for air and methane, respectively, are equal, R΄ of air = 287 J/kg K˚; R΄ methane = 520 J/kg K˚.
The last equation is sometimes written as:
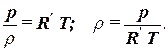 | (1.50) |
It can be seen from the last equation that the density of a gas depends on pressure and temperature, so if the density of a gas is known, then it is necessary to indicate the pressure, temperature, and composition of the gas, which is inconvenient. Therefore, the concepts of normal and standard physical conditions are introduced.
The pressure surface begins to slowly rise, and as you do so, you are not yet following this rise and your altimeter will indicate a descent. In true field you will start flying at higher pressure in this case as the 900mb surface rises above you and the anoderoid plate in your altimeter will indicate lower altitude and descent. However, you don't really realize this while flying, and will simply minimize vertical speed and maintain altitude, blissfully unaware that you are actually flying on an incline of constant pressure.
Normal conditions correspond to temperature t = 0°C and pressure p at = 0.1013°MPa. The density of air under normal conditions is equal to ρ v.n.us = 1.29 kg / m 3.
Standard Conditions correspond to temperature t = 20°C and pressure p at = 0.1013°MPa. The density of air under standard conditions is ρ w.st.us = 1.22 kg / m 3.
Therefore, from the known density under given conditions, it is possible to calculate the gas density at other values of pressure and temperature:
To better illustrate this, consider the following figure. In this picture, reds mean warmer than the average column of air, and blues means colder than the middle column. The whitish area in the middle represents the column at medium temperatures. Black solid lines are isobars. The black black line is the true height above the surface.
You should notice that the pressure levels in a warm column are farther apart because the air is less dense and it requires more energy. Likewise, in a cool column, the pressure levels are closer together because the air is denser than standard air.
Excluding reservoir temperature, we obtain the ideal gas equation of state, which we will use in the future:
where z - coefficient characterizing the degree of deviation of the state of a real gas from the law of ideal gases (supercompressibility coefficient) and depending for a given gas on pressure and temperature z = z(p, T) . Values of the coefficient of supercompressibility z are determined by the graphs of D. Brown.
To relate this to the discussion above, consider yourself in the standard column at true height above the ground, represented by the dotted line. Your altimeter does not sense this true altitude, but instead senses pressure outside the aircraft. This will be roughly calibrated to your true altitude, but using the local altimeter setting. Now, as you fly left or right and maintain a constant indicated altitude, you will track the thick line, as this is the pressure corresponding to your true altitude at standard pace.
Oil viscosity. Experiments show that the viscosity coefficients of oil (at pressures above saturation pressure) and gas increase with increasing pressure. With significant pressure changes (up to 100 MPa), the dependence of the viscosity of reservoir oils and natural gases on pressure can be taken exponential:
| | (1.56) |
For small changes in pressure, this dependence is linear.
When you fly into a colder column you will actually descend and you will climb when you fly into a warmer column. More compressed by excess air mass than at higher altitudes: so the air is denser. Air always has the highest density and pressure on earth - and the most high temperature except for inversions. At higher altitudes, the air gets thinner and thinner. If the temperature were the same at all altitudes, the air pressure and air density would also decrease with increasing altitude, in accordance with the gas law.
Here m0 – viscosity at fixed pressure p0 ; β m - coefficient determined experimentally and depending on the composition of oil or gas.
Formation porosity. To find out how the porosity coefficient depends on pressure, consider the question of stresses acting in a porous medium filled with liquid. When the pressure in the liquid decreases, the forces on the skeleton of the porous medium increase, so the porosity decreases.
However, the temperature at different altitudes varies greatly. 90% of the atmosphere is below 20 km. 70% of the atmosphere is below 10 km above sea level. 55% of the atmosphere is below 5 km above sea level. As you can see, these variables are highly dependent on temperature.
Accurate determination of air density
Determining the exact density of air requires accounting for humidity, as this changes the gas constant of the air. After adjusting the gas constant, Eq. The gas constant of humid air is calculated using the formula. Saturated vapor pressure, note restrictions. The equation provides the vapor pressure in Pascal. Alternatively, formula.
Due to the small deformation of the solid phase, it is usually considered that the change in porosity depends linearly on the change in pressure. The rock compressibility law is written as follows, introducing formation volumetric elasticity coefficient b c:
where m0 – coefficient of porosity at pressure p0 .
Laboratory experiments for various granular rocks and field studies show that the coefficient of volumetric elasticity of the formation is (0.3 - 2) 10 -10 Pa -1 .
Use; particularly precise values are provided by the panel. The atmosphere is the layer of air that surrounds our planet. Other planets in solar system also have atmosphere. The gases that make up the atmosphere are held around the Earth due to the attraction of gravity and accompany its movement.
Air density decreases as altitude increases, with 50% of gases and particles in suspension located in the first 5 km. The atmosphere is fundamental to sustaining life on Earth because. It is a source of oxygen, a necessary gas for life, which regulates the temperature and the earth's climate, which is responsible for the distribution of water on the planet, protects the Earth from cosmic radiation and meteors. Atmosphere: Our protective shield.
With significant changes in pressure, the change in porosity is described by the equation:
and for large - exponential:
| | (1.61) |
In fractured reservoirs, the permeability changes more intensively depending on pressure than in porous ones; therefore, in fractured reservoirs, taking into account the dependence k(p) more necessary than in granular.
The Earth's atmosphere presents various characteristics along its vertical profile and is approximately 000 km thick. The column of air that creates it exerts a pressure called atmospheric pressure. Because it depends on the density of the air, as we ascend, the atmospheric pressure becomes less.
Atmospheric pressure also varies along the earth's surface, which is an important variable for meteorological analysis. The atmosphere is also responsible for observing blue skies during the day, as blue sky particles scatter predominantly visible radiation at that wavelength.
The equations of state of the liquid or gas saturating the formation and the porous medium complete the system of differential equations.
Abstract on the topic:
Air density
Plan:
- Introduction
- 1
Relationships within the ideal gas model
- 1.1 Temperature, pressure and density
- 1.2 Influence of air humidity
- 1.3 Influence of height above sea level in the troposphere
Notes
Introduction
Air density- the mass of gas of the Earth's atmosphere per unit volume or the specific mass of air under natural conditions. Value air density is a function of the height of the measurements taken, of its temperature and humidity. Usually the standard value is considered to be 1.225 kg ⁄ m 3 , which corresponds to the density of dry air at 15°C at sea level.
Due to the different characteristics that the atmosphere presents, it is divided into layers at different altitudes. The layer closest to the Earth's surface is called the troposphere. It extends up to an average height of 12 km. This layer corresponds to 80% of the total mass of the atmosphere and is where the main meteorological phenomena occur. The temperature decreases with altitude.
The greenhouse effect is a natural and essential phenomenon for living beings. This prevents the Earth from losing too much heat, resulting in extreme temperature fluctuations. With the increase in greenhouse gas emissions from human activities, the rise in global temperature is being tested.
1. Relationships within the ideal gas model
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
1.1. Temperature, pressure and density
The density of dry air can be calculated using the Clapeyron equation for an ideal gas at a given temperature (English) Russian and pressure:
Air update environment can be classified as. Natural or spontaneous ventilation Dynamic ventilation Thermal ventilation. Positive pressure Negative pressure. . The amount of air that the ventilation system must bring in or remove from the aviary depends on weather conditions and the age of the birds.
Natural or spontaneous ventilation. This is the normal movement of air, which can occur due to differences in pressure caused by the action of wind or temperature between the two media in question. The cause of wind is the difference in atmospheric pressure at ground level, which in turn is a consequence of temperature changes.
Here ρ - air density, p- absolute pressure, R- specific gas constant for dry air (287.058 J ⁄ (kg K) ), T is the absolute temperature in Kelvin. So by substitution we get:
- under the standard atmosphere of the International Union of Pure and Applied Chemistry (temperature 0 ° C, pressure 100 kPa, zero humidity), air density is 1.2754 kg ⁄ m³;
- at 20 °C, 101.325 kPa and dry air, the density of the atmosphere is 1.2041 kg ⁄ m³.
The table below shows various air parameters calculated on the basis of the corresponding elementary formulas, depending on temperature (pressure is taken as 101.325 kPa)
Air always flows from a point high pressure to the point low pressure. This means that the air velocity in the installation is always greater in the windward side openings than in the leeward side. The action of the winds, though intermittent, causes a staggering pressure in the horizontal direction. When the air current loses speed, the pressure increases. The higher the pressure difference, the higher the air velocity.
Figure Pressure scaling in the horizontal direction. Dynamic ventilation is enhanced through openings conveniently located in opposite walls and in the direction of the prevailing winds. The rate at which natural ventilation occurs depends on the speed and direction of the wind, the proximity and size of obstacles such as mountains or buildings, and the shape and location of air inlets and outlets.
1.2. Influence of air humidity
Humidity refers to the presence of gaseous water vapor in the air, the partial pressure of which does not exceed the saturated vapor pressure for given atmospheric conditions. The addition of water vapor to air causes a decrease in its density, which is explained by the lower molar mass of water (18 g ⁄ mol) compared to the molar mass of dry air (29 g ⁄ mol). Humid air can be considered as a mixture of ideal gases, the combination of the densities of each of which makes it possible to obtain the required value for their mixture. This interpretation allows the determination of the density value with an error level of less than 0.2% in the temperature range from -10 °C to 50 °C and can be expressed as follows:
When the wind hits the enclosure, different areas of positive pressure and negative pressure can form. Positive pressure exceeding normal atmospheric pressure characterizes the movement of the air mass against the enclosure and the negative attraction of the air mass. Because air moves from points greater than that of lower pressure, if there are holes in the aviary, the positive pressure will cause a mass of air to enter through the holes and refuse to leave. It makes no sense to have holes in the same plane, since pressures that are equal do not lead to air circulation.
where is the density of moist air (kg ⁄ m³); p d- partial pressure of dry air (Pa); R d- universal gas constant for dry air (287.058 J ⁄ (kg K)); T- temperature (K); p v- water vapor pressure (Pa) and R v- universal constant for steam (461.495 J ⁄ (kg K) ). Water vapor pressure can be determined from relative humidity:
This means that vents must be on opposite walls for effective ventilation. This type of natural ventilation is known as "cross ventilation". With natural ventilation in an aviary, by opening the ridge and side openings, air flows from a high pressure point to a low pressure point. If the negative pressure on the ridge is greater than the negative pressure on the lee side, air flows from the latter to the open ridge.
In thermal ventilation, temperature differences cause changes in the density of the air in the enclosures, which causes the pressure to be directed in the vertical direction by an exhaust or thermosyphon. This pressure difference depends on the temperature difference between the air inside and outside the enclosure, the size of the holes for air inlet and outlet through the lantern, and, finally, the level difference between these holes. This effect is also referred to as the "chimney effect" and, given the natural ventilated enclosure, this effect exists regardless of the outside air velocity.
where p v- water vapor pressure; φ - relative humidity and p sat is the partial pressure of saturated vapor, the latter can be represented as the following simplified expression:
which gives the result in millibars. Dry air pressure p d determined by a simple difference:
where p denotes the absolute pressure of the system under consideration.
1.3. Influence of height above sea level in the troposphere
The dependence of pressure, temperature and air density on altitude compared to the standard atmosphere ( p 0 \u003d 101325 Pa, T0\u003d 288.15 K, ρ 0 \u003d 1.225 kg / m³).
The following parameters can be used to calculate the air density at a certain height in the troposphere (the value for the standard atmosphere is indicated in the atmospheric parameters):
- standard atmospheric pressure at sea level - p 0 = 101325 Pa;
- standard temperature at sea level - T0= 288.15K;
- acceleration of free fall over the surface of the Earth - g\u003d 9.80665 m ⁄ sec 2 (for these calculations it is considered a value independent of height);
- rate of temperature drop (English) Russian with height, within the troposphere - L= 0.0065 K ⁄ m;
- universal gas constant - R\u003d 8.31447 J ⁄ (Mol K) ;
- molar mass of dry air - M= 0.0289644 kg ⁄ Mol.
For the troposphere (i.e., the region of linear temperature decrease - this is the only property of the troposphere used here), the temperature at altitude h above sea level can be given by the formula:
pressure at altitude h:
Then the density can be calculated by substituting the temperature T and pressure P corresponding to a given height h into the formula:
These three formulas (dependence of temperature, pressure and density on height) are used to construct the graphs shown on the right. Graphs are normalized - they show the general behavior of the parameters. "Zero" values for correct calculations must each time be substituted in accordance with the readings of the relevant instruments (thermometer and barometer) at the moment at sea level.
